Nearly half of all Americans, or more than 155 million people, will live in one of the 39 states or Washington, D.C. where marijuana is legal for medicinal purposes by 2023.
To maintain a sufficient supply for the market as the demand for cannabis keeps rising, cultivation has increased. To assure the quality and safety of the products, this increase has forced the area of microbiology to make important improvements. Cannabis is a crop that requires a lot of upkeep and complicated agricultural factors.
It’s critical to project that a percentage of your product may fail compliance testing in the state where you operate if you are involved in any aspect of the seed-to-sale process. In the industry, failure rates generally vary from 10% to 20%.
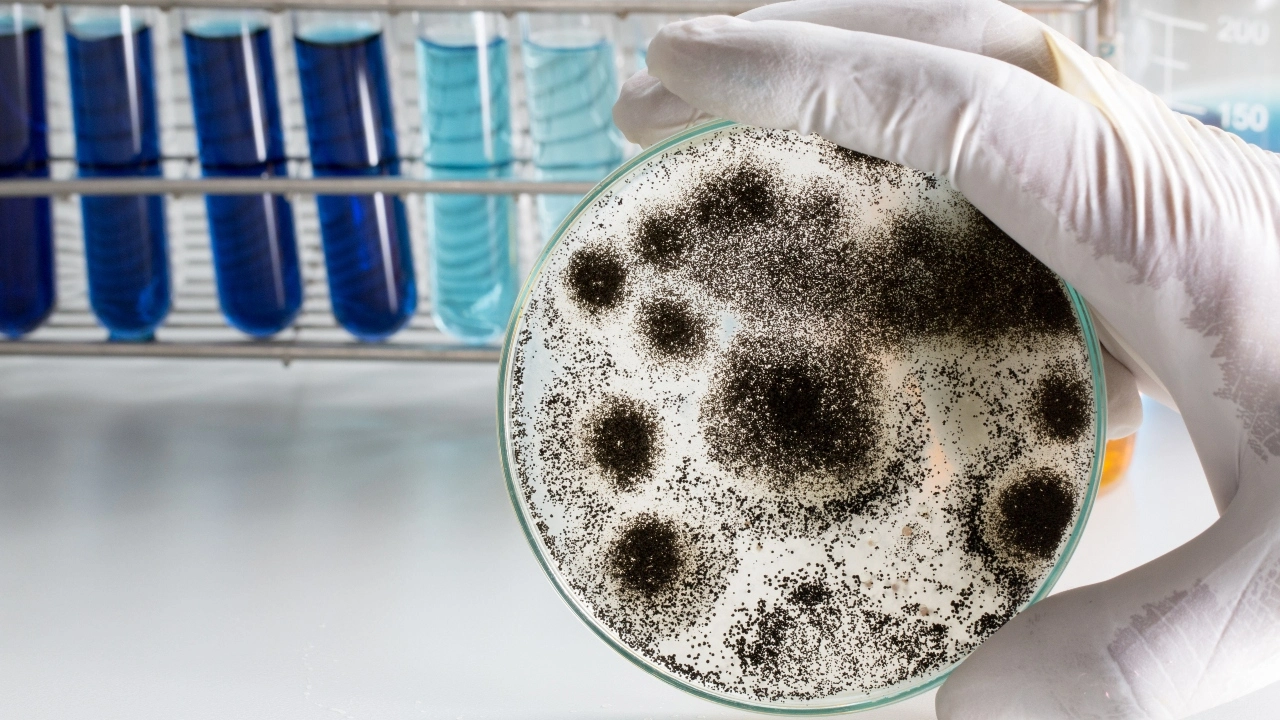
The presence of mould, particularly Aspergillus species, which are recognised human diseases, is the most frequent reason for failure. Aspergillus testing has increased significantly in recent months, raising worries that the results might produce false positives and casting doubt on the reliability of the testing procedures.
This claim about “false” good outcomes is untrue, in actuality. Despite the fact that more tests are being performed, diagnostic data shows that the typical positive identification rate for Aspergillus is between 10% and 20%.
Non-navigational testing for cannabis is mostly focused on Aspergillus moulds since their presence can endanger both immunocompromised people and medical patients, sometimes even fatalities. There are Aspergillus spores wherever you look. The genus has approximately 250 species and strains that have been discovered, and because they are numerous and quickly spread through the air, soil, and water, they are widely used in cannabis cultivation.
Most of the continuing talks in the cannabis sector centre on how gathered data may guarantee trust in testing procedures and findings while supplying a comprehensive picture of the production pipeline.
Storytelling with data is crucial. Parity with industry average failure rates may point to problems with testing procedures or serious failures during production.
You must make sure that your testing procedure is appropriate for your goals and customised for your particular laboratory, otherwise you risk getting inconsistent findings.
To achieve trustworthy third-party validation, the collaborative approach should include access to extensive data that identifies the distinctiveness of the technique or research objectives.
Sharing a common understanding with your testing partners and developers facilitates the process of identifying and separating objectives from non-objectives. This enables exact customisation that has to be incorporated.
In this instance, in silico analysis refers to the comparison of existing databases of bacterial or fungal DNA with technique objectives simulated on a computer.
You don’t have to be an expert in science or microbiology to comprehend the collaborative review process. When you are still in the brainstorming phase, you may question your testing partner directly about the collaboration data specific to your situation.

Cooperative reviews are helpful in enhancing compliance within your current process pipeline, particularly when an environmental field trip is being planned to highlight your particular findings.
You should be prepared for the normal results of an Aspergillus test and confident in your testing strategy.
It is preferable to treat your Aspergillus tests as true positives due to the nature of this fungus.
Regular data assessment is the most efficient technique to lower your failure rate and create proactive measures to eliminate process flaws.